ROCHESTER, N.Y., Aug. 14, 2024 (GLOBE NEWSWIRE) -- Vaccinex (Nasdaq: VCNX) today provided an update on new clinical findings from its SIGNAL-AD Phase 1b/2 trial of pepinemab antibody in Alzheimer’s disease.
Our Company recently announced positive results of the phase 1b/2 study of its lead product, pepinemab, in early stages of Alzheimer’s disease (AD). The purpose of this report is to share additional data related to cognitive effects that may help clarify the goals of this study, how well it succeeded, and how this success supports continued development of pepinemab in AD and other neurodegenerative diseases including Huntington’s Disease (HD), which was the focus of a larger previously completed phase 2 study.
Following completion of its phase 2 study in HD, the Company recognized that a major market and strategic focus of its potential pharmaceutical partners was AD. The Company, therefore, undertook to determine whether pepinemab had similar effects in AD as it had reported for HD. The SIGNAL-AD clinical trial was a smaller, cost-effective study in AD that focused on common features of disease progression in AD and HD, including an important efficacy endpoint, the well-characterized decline in brain metabolic activity reflected by FDG-PET signal in a key brain region that is affected early in disease. For AD, one such region, identified in multiple prior studies, is the medial temporal cortex from which disease manifestations spread to other temporal regions and, over time, to the rest of the brain. In the randomized SIGNAL-AD study, that goal was clearly reached by data showing a statistically significant difference (p=0.0297) in FDG-PET signal in the medial temporal cortex following 12-months of treatment with pepinemab compared to placebo. Pepinemab was well-tolerated in AD, consistent with prior clinical experience in HD.
Scientific and clinical background:
Are there any other markers of pepinemab activity in AD and HD?
Yes. Additional secondary endpoints of the SIGNAL and SIGNAL-AD trials included blood-based biomarkers of astrocyte reactivity (inflammation) and neurodegeneration, as well as clinically meaningful measures of cognitive decline. In an extended series of prior studies, we observed that SEMA4D is upregulated in neurons during progression of both AD and HD. We also reported that astrocytes are the main cells in the brain that express high affinity plexin receptors for SEMA4D and, when SEMA4D binds to these receptors, it triggers dramatic transformation of astrocytes from the normal supportive physiological state to a reactive state associated with neuroinflammation. In their reactive state, astrocytes release a characteristic marker, GFAP, that can be detected in blood. A general model for progression of neurodegenerative disease is that some initial abnormality in brain, such as formation of toxic aggregates of Aβ amyloid in AD or of mutant huntingtin protein in HD, triggers astrocyte reactivity, which leads to neuronal damage and synaptic loss followed by cognitive decline. In AD in particular, neuronal damage is associated with formation of “tau tangles,” aggregates of the tau protein that are thought to accumulate as a result of disease associated processes and are toxic to neurons. A marker of such “tau-pathology” and neuronal damage is the release of a peptide, p-tau 217, into blood. In the SIGNAL-AD study, we employed a very sensitive assay to detect changes in concentration of both GFAP and p-tau 217 in blood samples of patients treated with pepinemab versus placebo.
Investors may recall that in our previously reported study of pepinemab treatment in HD, we focused on patients at a very early stage of progression as reflected by a Total Functional Capacity (TFC) score between 11 and 13, which is the top of the 13-point range of this functional scale. As a result, we did not know whether pepinemab would be equally effective in patients with more advanced disease (e.g. TFC 7-10). It was, therefore, important to assess the impact of pepinemab treatment in both earlier and later stages of disease in our study of AD. We accordingly enrolled equal numbers of AD patients with Mild Cognitive Impairment (MCI) and those who had progressed to the later stage of early dementia.
An important initial observation in the SIGNAL-AD trial was that changes in the levels of GFAP and pTau-217 biomarkers in blood correlated over 12-months of treatment, with a positive relationship between the two measures (correlation coefficient >0.5), and a highly significant p-value (p< 0.001). This supports the connection between astrocyte reactivity and neuronal damage. We also recently reported at the Alzheimer’s Association International Conference in Philadelphia, July 28 to August 1, 2024, that the increase in level of GFAP in blood that is characteristic of patients with MCI due to AD is reduced by treatment with pepinemab, and that there appears to be a similar effect of pepinemab treatment on p-tau 217 levels in patients with MCI. It was, however, striking that there was no discernible effect of treatment on either GFAP or p-tau 217 levels in blood of patients whose disease had advanced to the later stage of early dementia. Importantly, this was consistent with our previously reported statistically significant effect on GFAP levels in the much larger study of patients with early stage HD that enrolled approximately 90 patients/study arm (p=0.04).
Is there a cognitive benefit to treatment with pepinemab?
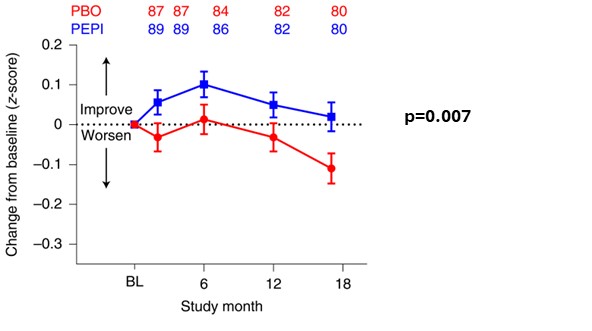
Cognitive decline is the major symptom of AD and is also cited by patients with HD and their families as their major concern during disease progression. Several different measures have been employed to assess cognitive change in AD. In Figures 1 (A) and (B), we show results from the SIGNAL-AD trial for the iADRS combination assessment of cognition introduced by Eli Lilly, and, in Figures 1 (C) and (D), we show results for a separate cognitive assessment, ADAS-Cog 13. In each case, we contrast the effect of pepinemab treatment in patients with MCI due to AD (Figures 1(A) and (C)) with for the apparent absence of an effect in patients diagnosed with early dementia (Figures 1(B) and (D)). We believe the evident trend of cognitive benefit from pepinemab treatment in patients with MCI is again strongly supported by statistically significant results from the previous larger study of patients with early stage HD (p=0.007), as shown in Figure 2. We plan to report further details and outcomes of the SIGNAL-AD trial at upcoming scientific conferences, as well as to publish a complete study report in a medical journal.
Figure 1. Pepinemab treatment appears to slow Cognitive decline in MCI due to AD
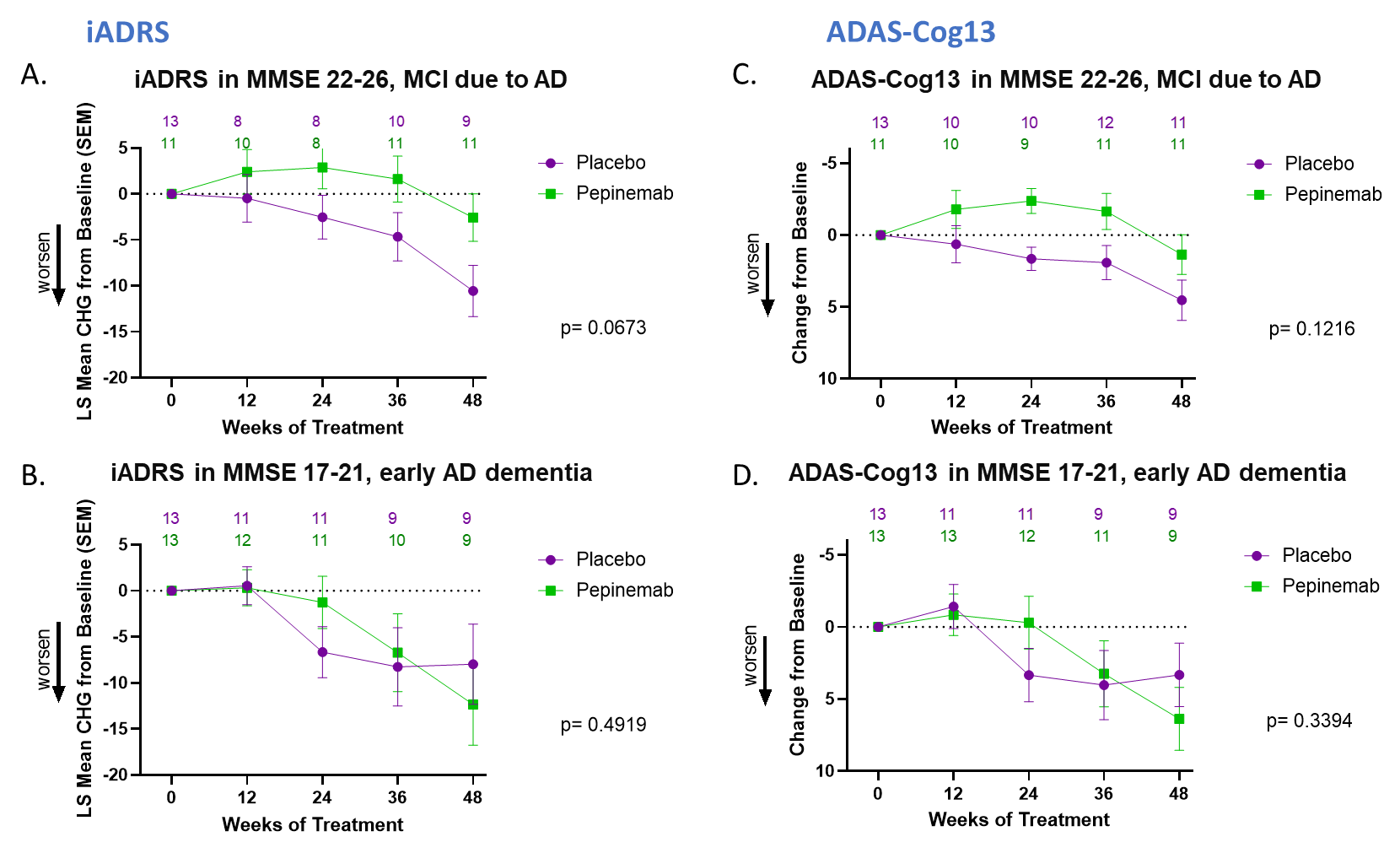
Figure 2. Pepinemab treatment appears to slow Cognitive decline in Huntington’s disease
Huntington’s Disease Cognitive Assessment Battery
Business Considerations
Is the benefit of pepinemab treatment in MCI due to AD clinically important, and what is the size of the population that might benefit from this treatment?
We believe that we have shown that pepinemab treatment is of benefit to patients with MCI due to AD, as was previously reported with statistical significance for patients with early stage HD, but that the benefit for patients with AD is lost or reduced during subsequent progression to dementia. It is, therefore, important to identify patients with MCI as early as possible and to treat to delay progression to dementia for as long as possible. We believe a drug that can slow or halt progression from MCI to dementia represents, at the present time, the most promising potential treatment option for AD. MCI is estimated to affect 1 million people with AD in the US alone, a very substantial population with an important unmet need.
The Company believes that it has compiled compelling early phase evidence of favorable tolerability and efficacy of pepinemab in neurodegenerative disease and that it is well-positioned to enter into a major partnership for continued development without a need for it to perform additional proof of concept studies. This partnership strategy would greatly reduce the Company’s operating expenses going forward.
The Company is very appreciative of past support by investors who enabled the progress we have reported.
With gratitude and confidence,
Maurice Zauderer, PhD
President & CEO, Vaccinex, Inc.
Forward Looking Statements
To the extent that statements contained in this letter are not descriptions of historical facts regarding Vaccinex, Inc. (“Vaccinex,” “we,” “us,” or “our”), they are forward-looking statements reflecting management’s current beliefs and expectations. Such statements include, but are not limited to, statements about the use and potential benefits of pepinemab treatment in patients with AD and HD; the potential and prospects for continuing late stage development of pepinemab, including as part of a prospective partnership; and other statements identified by words such as “believe,” “being,” “will,” “appear,” “expect,” “ongoing,” “potential,” “promising,” “indicate,” “suggest,” “apparent”, and similar expressions or their negatives (as well as other words and expressions referencing future events, conditions, or circumstances). Forward-looking statements involve substantial risks and uncertainties that could cause the outcome of our research and pre-clinical development programs, clinical development programs, future results, performance, or achievements to differ significantly from those expressed or implied by the forward-looking statements. Such risks and uncertainties include, among others, uncertainties inherent in the execution, cost and completion of preclinical studies and clinical trials, risks related to reliance on third parties, that interim and preliminary data may not be predictive of final results and does not ensure success in later clinical trials, uncertainties related to regulatory approval, risks related to our dependence on our lead product candidate pepinemab, the possible delisting of our common stock from Nasdaq if the Company is unable to regain and sustain compliance with the Nasdaq listing standards, and other matters that could affect our development plans or the commercial potential of our product candidates. Except as required by law, the Company assumes no obligation to update these forward-looking statements. For a further discussion of these and other factors that could cause future results to differ materially from any forward-looking statement, see the section titled “Risk Factors” in our periodic reports filed with the Securities and Exchange Commission and the other risks and uncertainties described in the Company’s annual year-end Form 10-K and subsequent filings with the SEC.
Investor Contact
Elizabeth Evans, PhD
Chief Operating Officer, Vaccinex, Inc.
(585) 271-2700
eevans@vaccinex.com
Photos accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/168ff1d7-5fbf-479c-87ba-fdde5d1e7e7c
https://www.globenewswire.com/NewsRoom/AttachmentNg/c663e516-9617-4f6e-bd88-37ce3940ec9c














